A) s®p
B) s®p*
C) n ®p*
D) p®p*
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the appropriate term for the mechanism of the addition of HBr to 1,3-dienes?
A) Nucleophilic addition
B) Electrophilic addition
C) Free radical addition
D) Conjugate addition
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an example of a bridged bicyclic system? 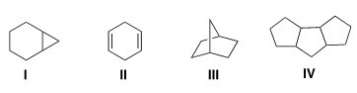
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following dienophile in order of increasing reactivity in a Diels-Alder reaction,listing the least reactive first. 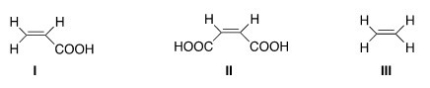
A) I < II < III
B) III < I < II
C) II < I < III
D) III < II < I
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the most reactive diene in a Diels-Alder reaction? 
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a major resonance contributor to the first structure? 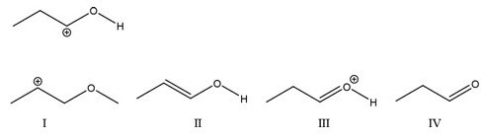
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of increasing stability,putting the least stable first. 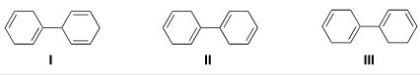
A) I < II < III
B) III < II < I
C) II < I < III
D) III < I < II
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 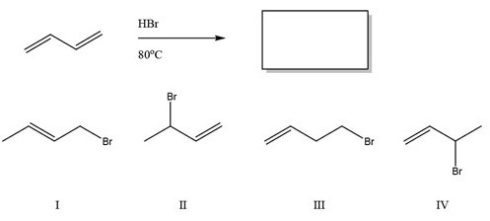
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 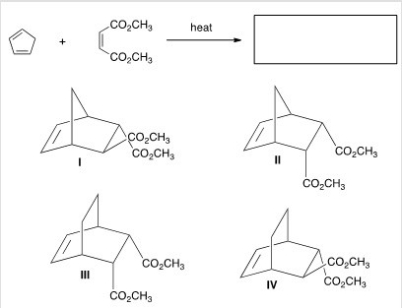
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the least reactive diene in a Diels-Alder reaction? 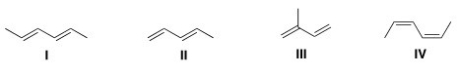
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 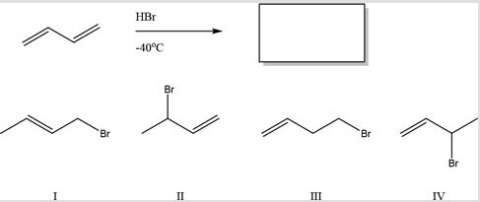
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is (are) the major product(s) of the following reaction? 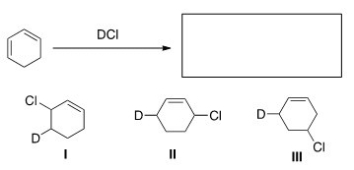
A) Only I
B) Only II
C) Only I and II
D) I,II,and III
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What diene and dienophile are used in a Diels-Alder reaction to prepare the following compound? 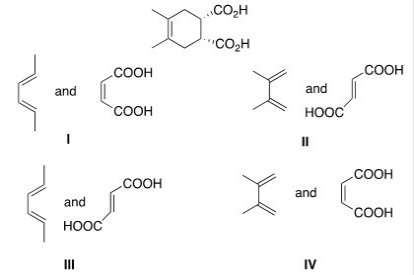
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is (are) conjugated dienes? 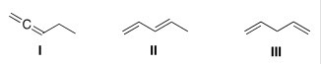
A) Only I
B) Only II
C) Only I and II
D) I,II,and III
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which triene has the largest heat of hydrogenation? 
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Which of the following statements about the Diels-Alder reaction is true?
A) The diene can react only when it adopts the s-trans conformation.
B) Electron-withdrawing substituents in the diene increase reaction rate.
C) Electron-donating substituents in the dienophile increase the reaction rate.
D) The stereochemistry of the dienophile is retained in the product.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not a feature of the Diels-Alder reaction?
A) They are initiated by peroxides.
B) They form new six-membered rings.
C) Three π bonds break.
D) They are concerted.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the kinetic product obtained from the addition of 1 equivalent of HBr to 1,3-butadiene?
A) 3-Bromo-1-butene
B) 1-Bromo-2-butene
C) 2-Bromo-2-butene
D) 2-Bromo-1-butene
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which triene absorbs the longest wavelength of UV light? 
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Which of the following is not a product obtained from the addition of 1 equivalent of HBr to (E) -1,3-pentadiene?
A) (E) -4-Bromo-2-pentene
B) (E) -1-Bromo-2-pentene
C) 3-Bromo-1-pentene
D) (E) -3-Bromo-2-pentene
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 53
Related Exams