A) Protons only
B) Electrons only
C) Positrons only
D) Neutrons only
E) Protons and Neutrons
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The radioisotope 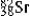 will decay through
will decay through
A) α decay.
B) β decay.
C) γ decay.
D) electron capture.
E) spontaneous fission.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following materials is put into a nuclear reactor to slow the chain reaction?
A) Heavy water
B) Moderators
C) Control rods
D) Reflectors
E) Chlorine
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following cannot be an example of nuclear fusion?
A) A nuclear reaction in which two reactants have greater binding energy than their fused product.
B) A nuclear reaction in which a carbon nucleus is produced.
C) The primary energy-producing reaction in the Sun.
D) The primary energy-producing reaction in a hydrogen bomb.
E) A reaction in which the reactants are less stable than the products.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Radioactive decay follows zero-order kinetics.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotope 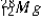 has a half-life of 21 hours. If a sample initially contains exactly 10000 atoms of
has a half-life of 21 hours. If a sample initially contains exactly 10000 atoms of  , approximately how many of these atoms will remain after one week?
, approximately how many of these atoms will remain after one week?
A) 1250
B) 78
C) 39
D) 0
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following series of radioactive decays would convert Pa-234 to Ra-226?
A) Beta, Alpha, Beta
B) Alpha, Alpha
C) Beta, Alpha, Alpha, Beta
D) Beta, Alpha, Alpha
E) Alpha, Beta, Gamma
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In living organisms, C-14 atoms disintegrate at a rate of 15.3 atoms per minute per gram of carbon. A charcoal sample from an archaeological site has a C-14 disintegration rate of 9.16 atoms per minute per gram of carbon. Estimate the age of this sample. The half-life of C-14 is 5730 years.
A) 3170 years
B) 3430 years
C) 4020 years
D) 4790 years
E) 6750 years
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
All the disintegrations of a sample of an unknown nuclide weighing 4.6 × 10-2 g were counted. In the first half-life of the sample, the total number of disintegrations counted was 4.3 × 1020. What is the atomic weight of the unknown element?
A) 32 amu
B) 16 amu
C) 8 amu
D) 4 amu
E) None of these choices are correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A patient's thyroid gland is to be exposed to an average of 5.5 µCi for 16 days as an ingested sample of iodine-131 decays. If the energy of the β radiation is 9.7 × 10-14 J and the mass of the thyroid is 32.0 g, what is the dose received by the patient?
A) 0.027 rads
B) 1.2 rads
C) 37 rads
D) 85 rads
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The s-process involves a slow succession of neutron absorption and beta decay processes during the normal life of a star.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotope 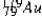 has a half-life of 7.5 seconds. If a sample contains 144 atoms of
has a half-life of 7.5 seconds. If a sample contains 144 atoms of 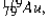 approximately how many such atoms were there present 30 seconds earlier?
approximately how many such atoms were there present 30 seconds earlier?
A) 576
B) 1152
C) 2304
D) 4320
E) 4.30 × 10 8
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An 85-kg person exposed to barium-141 receives 2.5 × 105 β particles, each with an energy of 5.2 × 10-13 J. How many rads does the person receive?
A) 2.4 × 10 -20
B) 1.5 × 10 -7
C) 1.8 × 10 -16
D) 6.1 × 10 -15
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotope 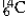 is unstable. This is predictable because
is unstable. This is predictable because
A) N/ Z ≠ 1.
B) N/ Z is relatively low and Z < 20.
C) N/ Z is relatively large and Z < 20.
D) Z is small.
E) N is large.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The radioisotope 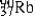 will decay through
will decay through
A) α decay.
B) β decay.
C) positron decay.
D) electron capture.
E) γ decay.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The radiochemist, Will I. Glow, studied thorium-232 and found that 2.82 × 10-7 moles emitted 8.42 × 106 α particles in one year. What is the decay constant for thorium-232?
A) 3.35 × 10 -14 yr -1
B) 4.96 × 10 -11 yr -1
C) 1.40 × 10 10 yr -1
D) 2.99 × 10 13 yr -1
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A scintillation counter
A) measures the signal coming from an ionized gas.
B) measures light emissions from excited atoms.
C) depends on an avalanche of electrons generated as a particle moves through a tube of argon gas.
D) detects high energy radiation better than low energy radiation.
E) detects an electric current in a gas.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A pure sample of tritium, 3H, was prepared and sealed in a container for a number of years. Tritium undergoes β decay with a half-life of 12.32 years. How long has the container been sealed if analysis of the contents shows there are 5.25 mol of 3H and 6.35 mol of 3He present?
A) 2.34 y
B) 3.38 y
C) 9.77 y
D) 14.1 y
E) 25.6 y
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Iodine-131, t1/2 = 8.0 days, is used in diagnosis and treatment of thyroid gland diseases. If a laboratory sample of iodine-131 initially emits 9.95 × 1018 β particles per day, how long will it take for the activity to drop to 6.22 × 1017 β particles per day?
A) 2.0 days
B) 16 days
C) 32 days
D) 128 days
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 7.85 × 10-5 mol sample of copper-61 emits 1.47 × 1019 positrons in 90.0 minutes. What is the decay constant for copper-61?
A) 0.00230 h -1
B) 0.00346 h -1
C) 0.207 h -1
D) 0.311 h -1
E) None of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 82
Related Exams