A) is greater than 7.0.
B) is equal to 7.0.
C) is less than 7.0.
D) is determined by the sizes of K a and K b.
E) is no longer affected by addition of base.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is the best representation of the titration curve that will be obtained in the titration of a weak diprotic acid H2A (0.10 mol L−1) with a strong base of the same concentration? 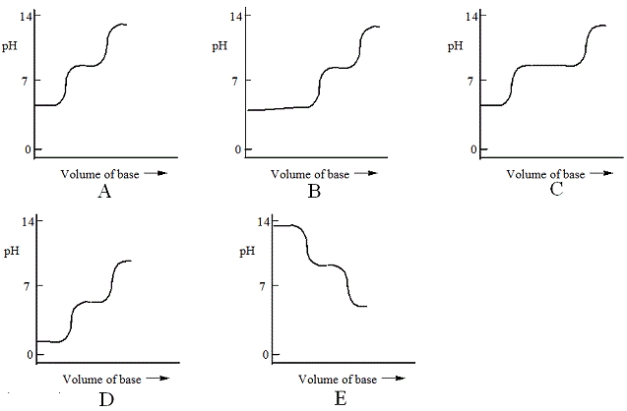
A) A
B) B
C) C
D) D
E) E
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
You need to prepare a buffer solution with a pH of 4.00, using NaF and HF. What ratio of the ratio of [base]/[acid] should be used in making the buffer? For HF, Ka = 7.2 × 10−4.
A) [base]/[acid] = 0.14
B) [base]/[acid] = 0.42
C) [base]/[acid] = 2.36
D) [base]/[acid] = 7.20
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of a monoprotic acid (HA) weighing 0.384 g is dissolved in water and the solution is titrated with aqueous NaOH. If 30.0 mL of 0.100 M NaOH is required to reach the equivalence point, what is the molar mass of HA?
A) 37.0 g/mol
B) 81.0 g/mol
C) 128 g/mol
D) 20.3 g/mol
E) 211 g/mol
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A popular buffer solution consists of carbonate (CO32−) and hydrogen carbonate (HCO3−) conjugate acid-base pair. Which, if any, of the following such buffers can neutralize the greatest amount of added hydrochloric acid, while remaining within its buffer range?
A) 1 L of 0.9 M CO 3 2− and 0.1 M HCO 3 −
B) 1 L of 0.1 M CO 3 2− and 0.9 M HCO 3 −
C) 1 L of 0.5 M CO 3 2− and 0.5 M HCO 3 −
D) 1 L of 0.1 M CO32− and 0.1 M HCO3−
E) They can all neutralize the same amount of hydrochloric acid.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 50.0-mL sample of 0.50 M HCl is titrated with 0.50 M NaOH. What is the pH of the solution after 28.0 mL of NaOH have been added to the acid?
A) 0.85
B) 0.75
C) 0.66
D) 0.49
E) 3.8
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the maximum amount of sodium sulfate that can be added to 1.00 L of 0.0020 M Ca(NO3) 2 before precipitation of calcium sulfate begins? Ksp = 2.4 × 10−5 for calcium sulfate.
A) 1.2 × 10 −2 mol
B) 4.9 × 10 −3 mol
C) 3.5 × 10 −3 mol
D) 1.2 × 10 −5 mol
E) 4.8 × 10 −8 mol
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The salts X(NO3) 2 and Y(NO3) 2 (where X+ and Y+ are metal ions) are dissolved in water to give a solution which is 0.1 M in each of them. Using the Ksp values listed below, decide which aqueous reagent, if any, will definitely precipitate X+ before precipitating Y+ from solution. Given Ksp values:XCl2, 1 × 10−5 YCl2, 1 × 10−10 X(OH) 2, 1 × 10−10 Y(OH) 2, 1 × 10−5
A) 1 M NH 3
B) 1 M HCl
C) 1 M HNO 3
D) 1 M NaCl
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The salts X(NO3) 2 and Y(NO3) 2 (where X+ and Y+ are metal ions) are dissolved in water to give a solution which is 0.1 M in each of them. Using the Ksp values listed below, decide which aqueous reagent, if any, will definitely precipitate X+ before precipitating Y+ from solution. Given Ksp values:XCl2, 1 × 10−5 YCl2, 1 × 10−10 X(OH) 2, 1 × 10−10 Y(OH) 2, 1 × 10−5
A) 1 M NaCl
B) 1 M HCl
C) 1 M HNO 3
D) 1 M NaOH
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the [H3O+] in a solution that consists of 1.2 M HClO and 2.3 M NaClO? Ka = 3.5 × 10−8
A) 7.8 × 10 −9 M
B) 1.8 × 10 −8 M
C) 6.7 × 10 −8 M
D) 1.6 × 10 −7 M
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The salts X(NO3) 2 and Y(NO3) 2 (where X+ and Y+ are metal ions) are dissolved in water to give a solution which is 0.1 M in each of them. Using the Ksp values listed below, decide which aqueous reagent, if any, will definitely precipitate X+ before precipitating Y+ from solution. Given Ksp values:XCl2, 1 × 10−5 YCl2, 1 × 10−10 X(OH) 2, 1 × 10−10 Y(OH) 2, 1 × 10−5
A) 1 M NaNO 3
B) 1 M HCl
C) 1 M HNO 3
D) 1 M NaCl
E) None of these choices are correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The solubility of lead(II) chloride is 0.45 g/100 mL of solution. What is the Ksp of PbCl2?
A) 4.9 × 10 −2
B) 1.7 × 10 −5
C) 8.5 × 10 −6
D) 4.2 × 10 −6
E) < 1.0 × 10 −6
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write the ion product expression for magnesium fluoride, MgF2.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the [H3O+] in a buffer that consists of 0.30 M HCOOH and 0.20 M HCOONa? For HCOOH, Ka = 1.7 × 10−4
A) 1.1 × 10 −4 M
B) 2.6 × 10 −4 M
C) 4.3 × 10 −4 M
D) 6.7 × 10 −5 M
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 114 of 114
Related Exams