A) 1, 2
B) 1, 3
C) 3, 4
D) 3
E) 2, 4
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
ΔSuniv = -1 for a spontaneous reaction.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The temperature at which the following process reaches equilibrium at 1.0 atm is the normal boiling point of hydrogen peroxide. (R = 8.314 J/K • mol) H2O2(l)  H2O2(g)
Use the following thermodynamic information at 298 K to estimate this temperature.
H2O2(g)
Use the following thermodynamic information at 298 K to estimate this temperature. 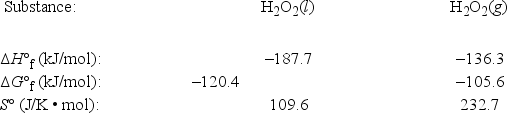
A) 120°C
B) 144°C
C) 196°C
D) 418°C
E) 585°C
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is necessary for a process to be spontaneous?
A) ΔHsys < 0
B) ΔSsys > 0
C) ΔSsurr < 0
D) ΔSuniv > 0
E) ΔGsys = 0
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a sky diver free-falls through the air, the process is
A) nonspontaneous because he is accelerating due to the force applied by gravity.
B) nonspontaneous because he is losing potential energy.
C) nonspontaneous because he had planned the jump for two weeks.
D) spontaneous.
E) in equilibrium.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate ΔG°rxn for the following reaction of ammonia with fluorine. 2NH3(g) + 5F2(g) → N2F4(g) + 6HF(g) 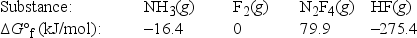
A) 179.1 kJ/mol
B) -179.1 kJ/mol
C) 1539.7 kJ/mol
D) -1539.7 kJ/mol
E) -211.9 kJ/mol
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is always true for an exothermic process?
A) qsys > 0, ΔSsurr < 0
B) qsys < 0, ΔSsurr > 0
C) qsys < 0, ΔSsurr < 0
D) qsys > 0, ΔSsurr > 0
E) w < 0
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 107 of 107
Related Exams