A) 0
B) 2
C) 4
D) 5
E) 6
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
If n = 2 then l = 0, -1, and 1.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Electrons will not pair in degenerate orbitals if an empty orbital is available and, according to Hund's rule, the degenerate orbitals must all contain one electron before any of them can contain two electrons.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm? (c = 3.00 × 108 m/s; h = 6.63 × 10-34 J • s; NA = 6.022 × 1023 moles-1)
A) 6.46 × 10-16 J
B) 6.46 × 10-25 J
C) 2.46 × 10-4 J
D) 12.4 kJ
E) 246 kJ
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What element is represented by the electron configuration 1s22s22p63s23p64s23d104p65s14d10?
A) Ag
B) Rb
C) Cd
D) Sr
E) Cu
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type of configuration specifies the arrangement of electrons in the orbitals of an atom?
A) Heisenberg configuration
B) Nuclear configuration
C) Atomic configuration
D) Proton configuration
E) Electron configuration
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
The following combination of quantum numbers is not allowed. Correct this set of quantum numbers by changing only one quantum number, and write in an appropriate corrected value. n = 2, l = 2, ml = 2; Corrected: ________ = _____
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the energy, in joules, required to excite a hydrogen atom by causing an electronic transition from the n = 1 to the n = 4 principal energy level. Recall that the energy levels of the H atom are given by En = -2.18 × 10-18 J(1/n2) .
A) 2.07 × 10-29 J
B) 2.19 × 105 J
C) 2.04 × 10-18 J
D) 3.27 × 10-17 J
E) 2.25 × 10-18 J
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 6.0-gram champagne cork is shot out of an opened champagne bottle and traveled at a speed of 65 km/hour. Calculate the de Broglie wavelength (in meters) of the cork.
A) 6.1 × 10-33 m
B) 1.7 × 10-36 m
C) 6.1 × 10-36 m
D) 1.7 × 10-33 m
E) 5.5 × 10-20 m
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ________ quantum number is the quantum number which describes the orientation of the orbital in space.
A) principal
B) angular momentum
C) magnetic
D) spin
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The orbital diagram for a ground-state nitrogen atom is 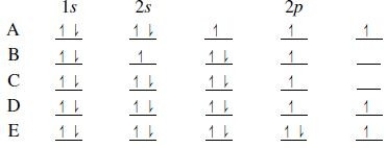
A) A
B) B
C) C
D) D
E) E
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a correct set of quantum numbers for an electron in a 3d orbital?
A) n = 3, l = 0, ml = -1
B) n = 3, l = 1, ml = +3
C) n = 3, l = 2, ml = 3
D) n = 3, l = 3, ml = +2
E) n = 3, l = 2, ml = -2
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the electron configuration for tungsten?
A) 1s22s22p63s23p64s23d104p65s24d105p66s24f145d6
B) 1s22s22p63s23p64s23d104p65s24d105p66s14f145d5
C) 1s22s22p63s23p64s23d104p65s24d105p66s24f145d4
D) 1s22s22p63s23p64s23d104p65s24d105p66s24f145d7
E) 1s22s22p63s23p54s23d104p65s24d105p66s24f145d7
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the wavelength of radiation that has a frequency of 6.912 × 1014 s-1? (c = 3.00 × 108 m/s)
A) 1.447 × 10-15 nm
B) 4.337 × 102 nm
C) 2.304 × 106 nm
D) 2.074 × 1023 nm
E) 4.337 × 10-7 nm
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the Rydberg equation to calculate the frequency of a photon absorbed when the hydrogen atom undergoes a transition from n1 = 2 to n2 = 4. (R = 1.096776 × 107 m-1; c = 3.00 × 108 m/s)
A) 2.06 × 106 s-1
B) 2.74 × 106 s-1
C) 6.17 × 1014 s-1
D) 8.23 × 1014 s-1
E) > 1015 s-1
G) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 121 - 135 of 135
Related Exams