A) Li2+, O-, H-
B) Li+, OH-
C) LiO-, H+
D) Li+, O2-, H+
E) Li-, OH+
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is a weak base?
A) NH3
B) Ca(OH) 2
C) Ba(OH) 2
D) NaOH
E) CH3COOH
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 350-mL sample of 0.276 M HNO3 is partially neutralized by 125 mL of 0.120 M Ca(OH) 2. Find the concentration of nitric acid in the resulting solution.
A) 0.0429 M
B) 0.140 M
C) 0.172 M
D) 0.203 M
E) 0.773 M
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If aqueous solutions of Mg(C2H3O2) 2 and LiOH are mixed, which insoluble precipitate is formed?
A) LiC2H3O2
B) Li(C2H3O2) 2
C) MgOH
D) Mg(OH) 2
E) CH3OH
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 0.8838-g sample of an ionic compound containing bromide ions and an unknown metal cation is dissolved in water and treated with an excess of AgNO3. If 1.573 g of a AgBr precipitate forms, what is the percent by mass of Br in the original compound?
A) 62.13%
B) 75.72%
C) 56.19%
D) 42.55%
E) 37.87%
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calcium nitrate tetrahydrate dissolves in warm water to the extent of 266 g per 100. cm3. What is the concentration of nitrate ions in this solution?
A) 32.4 M
B) 22.5 M
C) 11.3 M
D) 16.2 M
E) 8.10 M
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What volume of a 0.442 M NaOH solution is needed to neutralize 65.0 mL of a 0.296 M solution of HNO3?
A) 8.71 mL
B) 21.8 mL
C) 43.5 mL
D) 87.1 mL
E) 174 mL
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these compounds is a strong electrolyte?
A) H2O
B) N2
C) CH3COOH (acetic acid)
D) C2H6O (ethanol)
E) KOH
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Sodium hydroxide is water soluble.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
A(n) ________ ________ is the name given for the substance that causes another substance to undergo oxidation.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If aqueous solutions of Na2CO3 and BaCl2 are mixed, which insoluble precipitate is formed?
A) Ba2CO3
B) BaCO3
C) NaCl
D) NaCl2
E) BaO
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the reducing agent in the following reaction? Mg + NiO2 + 2H2O → Mg(OH) 2 + Ni(OH) 2
A) Mg
B) NiO2
C) H2O
D) Mg(OH) 2
E) Ni(OH) 2
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which salt is produced by the neutralization of sodium hydroxide with sulfuric acid?
A) Na2SO4
B) Na2(SO4) 3
C) Na(SO4) 2
D) Na2SO3
E) Na2S
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the solubility rules, which of these processes will occur when solutions of ZnSO4(aq) and MgCl2(aq) are mixed?
A) ZnCl2 will precipitate; Mg2+ and SO42- are spectator ions.
B) ZnSO4 will precipitate; Mg2+ and Cl- are spectator ions.
C) MgSO4 will precipitate; Zn2+ and Cl- are spectator ions.
D) MgCl2 will precipitate; Zn2+ and SO42- are spectator ions.
E) No precipitate will form.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these compounds is a nonelectrolyte?
A) NaF
B) HNO3
C) CH3COOH (acetic acid)
D) NaOH
E) C6H12O6 (glucose)
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 50.0 mL sample of 0.436 M NH4NO3 is diluted with water to a total volume of 250.0 mL. What is the ammonium nitrate concentration in the resulting solution?
A) 2.18 M
B) 0.523 M
C) 0.349 M
D) 0.174 M
E) 0.0872 M
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is a strong base?
A) LiOH
B) CH3COOH
C) NH3
D) H3PO4
E) HClO2
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which accurately characterizes all nonelectrolyte solutions?
A) Nonelectrolyte solutions contain molecules.
B) Nonelectrolyte solutions conduct electricity.
C) Nonelectrolyte solutions react with other solutions.
D) Nonelectrolyte solutions do not conduct electricity.
E) Nonelectrolyte solutions conduct heat.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If aqueous solutions of ammonium sulfide and copper(II) nitrate are mixed, which insoluble precipitate is formed?
A) CuS
B) Cu2S
C) NH4NO3
D) NH4(NO3) 2
E) CuSO4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The diagram below represents the final mixture corresponding to which chemical process? (Each sphere represents 1 mole of ions.) 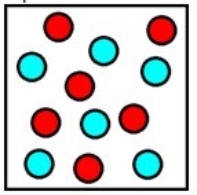
A) Six moles of sodium metal are added to six moles of hydrochloric acid.
B) Six moles of magnesium metal are added to six moles of hydrochloric acid.
C) Six moles of aluminum metal are added to twelve moles of hydrochloric acid.
D) Twelve moles of sodium metal are added to twelve moles of hydrochloric acid.
E) Twelve moles of magnesium metal are added to twelve moles of hydrochloric acid.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 146
Related Exams