Correct Answer

verified
L is a strong-field ligand. Reasoning (a...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
The ion [Co(NH3) 6]2+ is octahedral and high spin. This complex is
A) paramagnetic, with 1 unpaired electron.
B) paramagnetic, with 3 unpaired electrons.
C) paramagnetic, with 4 unpaired electrons.
D) paramagnetic, with 5 unpaired electrons.
E) diamagnetic.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The best name for K4[FeCl2(CN) 4] is
A) tetrapotassium dichlorodicyanoiron(II) .
B) potassium dichlorodicyanoiron(II) .
C) potassium dichlorodicyanoferrate(III) .
D) tetrapotassium dichlorobis(cyano) iron(III) .
E) potassium dichlorotetracyanoferrate(II) .
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many 3d electrons does a V3+ ion have?
A) 6
B) 5
C) 4
D) 3
E) 2
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Predict the number of unpaired electrons in the [Fe(CN)6]3- ion.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The numbers of geometrical isomers and optical isomers of the complex ion [Co(en) 3]3+ are, respectively,
A) 2 and 2.
B) 1 and 1.
C) 3 and 2.
D) 1 and 2.
E) 2 and 4.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Write the chemical formula of diamminedichloroplatinum(II).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many 3d electrons does a Mn2+ ion have?
A) 1
B) 2
C) 3
D) 4
E) 5
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electron configuration of a nickel atom is
A) [Ar]4s23d6.
B) [Ar]4s13d7.
C) [Ar]3d8.
D) [Ar]4s23d8.
E) [Ar]3d10.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the coordination compound K2[Co(en) Cl4], the coordination number (C.N.) and oxidation number (O.N.) of cobalt are
A) C.N. = 6; O.N. = +2.
B) C.N. = 6; O.N. = +3.
C) C.N. = 5; O.N. = +2.
D) C.N. = 5; O.N. = +4.
E) C.N. = 4; O.N. = +3.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the coordination number (C.N.) and oxidation number (O.N.) of the metal atom in the coordination compound [Cr(NH3) 2(en) Cl2].
A) C.N. = 4; O.N. = +2.
B) C.N. = 5; O.N. = +3.
C) C.N. = 5; O.N. = +2.
D) C.N. = 6; O.N. = +3.
E) C.N. = 6; O.N. = +2.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the complex ion [ML6]n+, Mn+ has seven d electrons and L is a strong field ligand. According to crystal field theory, the magnetic properties of the complex ion correspond to how many unpaired electrons?
A) 0
B) 1
C) 2
D) 3
E) 5
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Name the complex ion [Ni(CN)4]2-.
Correct Answer

verified
tetracyano...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
In the complex ion [ML6]n+, Mn+ has six d electrons and L is a weak field ligand. According to crystal field theory, the magnetic properties of the complex ion correspond to how many unpaired electrons?
A) 0
B) 1
C) 2
D) 3
E) 4
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electron configuration of a Co3+ ion is
A) [Ar]3d6.
B) [Ar]4s13d5.
C) [Ar] 4s23d4.
D) [Ar]3d5.
E) [Ar]3d4.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The coordination compound shown below has 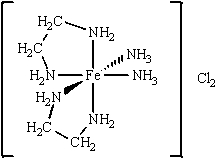
A) two monodentate ligands and two bidentate ligands.
B) three monodentate ligands and two bidentate ligands.
C) four monodentate ligands and two bidentate ligands.
D) one monodentate ligand and four bidentate ligands.
E) four bidentate ligands.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
What terms describe the geometric isomers that are possible for the complex [CrF2Cl4]3-?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these ligands produces the weakest crystal field?
A) CN-
B) I-
C) OH-
D) H2O
E) NH3
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electron configuration of a Cr3+ ion is
A) [Ar]3d5.
B) [Ar]4s13d2.
C) [Ar]3d3.
D) [Ar]4s13d5.
E) [Ar]4s23d4.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the coordination number of silver in [Ag(NH3)2]Cl?
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 76
Related Exams