Correct Answer

verified
The electr...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Write the ground state electron configuration for the phosphorus atom.
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the total number of electrons possible in the 6s orbital?
Correct Answer

verified
Correct Answer
verified
Short Answer
The orbital diagram for a ground-state nitrogen atom is 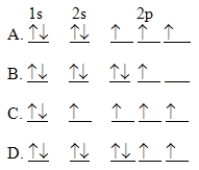
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the wavelength, in meters, of an alpha particle with a kinetic energy of 8.0 * 10-13 J.[mass of an alpha particle = 4.00150 amu; 1 amu = 1.67 *10-27 kg]
Correct Answer

verified
6.4 * 10View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
The electron configuration of a ground-state Co atom is
A) [Ar]4s23d7
B) 1s22s22p63s23d9
C) [Ne]3s23d7
D) [Ar]4s13d5
E) [Ar]4s24d7
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The Bohr model of the hydrogen atom found its greatest support in experimental work on the photoelectric effect.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Each shell (principal energy level)of quantum number n contains n subshells
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
A possible set of quantum numbers for the last electron added to complete an atom of gallium (Ga)in its ground state is 
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the electron configuration of an excited state of an oxygen atom?
A) 1s22s22p4
B) 1s22s22p5
C) 1s22s22p33s1
D) 1s22s22p6
E) 1s22s22p3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electron in a hydrogen atom falls from an excited energy level to the ground state in two steps, causing the emission of photons with wavelengths of 1870 and 102.5 nm. What is the quantum number of the initial excited energy level from which the electron falls?
A) 2
B) 3
C) 4
D) 6
E) 8
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the electron configuration of an excited state of a copper atom?
A) [Ar]4s23d9
B) [Ar]4s13d10
C) [Ar]4s13d8
D) [Ar]4s23d8
E) [Ar]4s03d10
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element has the following ground-state electron configuration? [Ar]4s23d104p5
A) aresnic
B) bromine
C) iodine
D) selenium
E) tellerium
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Write the ground state electron configuration for Cr.
Correct Answer

verified
Correct Answer
verified
Short Answer
The colors of the visible spectrum are blue, green, orange, red, violet, and yellow.Of these colors, ______ has the least energy.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element has the following ground-state electron configuration? 1s22s22p63s2
A) Na
B) Mg
C) Al
D) Si
E) Ne
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Write the ground state electron configuration for the selenium atom.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the maximum number of electrons in an atom that can have the following set of quantum numbers? n = 4 l = 3 ml = -2 ms = +1/2
A) 0
B) 1
C) 2
D) 6
E) 10
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many unpaired electrons does a ground-state atom of sulfur have?
A) 0
B) 1
C) 2
D) 3
E) 4
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Calculate the energy of a photon of light with a wavelength of 360 nm.
Correct Answer

verified
5.5 * 10View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 61 - 80 of 115
Related Exams