A) always positive
B) always negative
C) sometimes positive, sometimes negative
D) always zero
E) unpredictable
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
To produce maximum heat, an explosive compound should have ________ chemical bonds and decompose to molecule with ________ bonds.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A valid Lewis structure of ________ cannot be drawn without violating the octet rule.
A) ClF3
B) PCl3
C) SO3
D) CCl4
E) CO2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electron configuration [Kr]4d10 represents ________.
A) Ag+
B) Cd
C) Cd+
D) Ag2+
E) Sr2+
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A nonpolar bond will form between two ________ atoms of ________ electronegativity.
A) different, opposite
B) identical, different
C) different, different
D) similar, different
E) identical, equal
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a given arrangement of ions, the lattice energy increases as ionic radius ________ and as ionic charge ________.
A) decreases, increases
B) increases, decreases
C) increases, increases
D) decreases, decreases
E) This cannot be predicted.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The diagram below is the Born-Huber cycle for the formation of crystalline potassium fluoride. 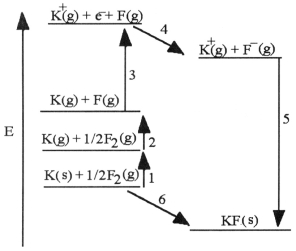 -Which energy change corresponds to the first ionization energy of potassium?
-Which energy change corresponds to the first ionization energy of potassium?
A) 2
B) 5
C) 4
D) 3
E) 6
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the table of average bond energies below, the △H for the reaction is ________ kJ. C≡O (g) + 2H2 (g) → H3C-O-H (g) Bond: C-O C=O C≡O C-H H-H O-H D (kJ/mol) : 358 799 1072 413 436 463
A) +276
B) -276
C) +735
D) -735
E) -116
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The diagram below is the Born-Huber cycle for the formation of crystalline potassium fluoride.  -Which energy change corresponds to the electron affinity of fluorine?
-Which energy change corresponds to the electron affinity of fluorine?
A) 2
B) 5
C) 4
D) 1
E) 6
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Bond enthalpy can be positive or negative.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Of the possible bonds between carbon atoms (single, double, and triple) , ________.
A) a triple bond is longer than a single bond
B) a double bond is stronger than a triple bond
C) a single bond is stronger than a triple bond
D) a double bond is longer than a triple bond
E) a single bond is stronger than a double bond
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Electropositivity ________ from left to right within a period and ________ from top to bottom within a group.
A) decreases, increases
B) increases, increases
C) increases, decreases
D) stays the same, increases
E) increases, stays the same
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the table of bond dissociation energies, the ΔH for the following gas-phase reaction is ________ kJ. 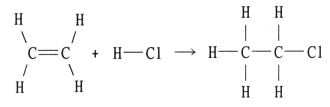
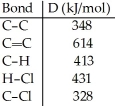
A) -44
B) 38
C) 304
D) 2134
E) -38
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The central atom in ________ does not violate the octet rule.
A) SF4
B) KrF2
C) CF4
D) XeF4
E) ICl4-
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The central atom in ________ violates the octet rule.
A) PF5
B) SF2
C) Cl2
D) Br2CO
E) FCN
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following does not have eight valence electrons?
A) Ca+
B) Rb+
C) Xe
D) Br-
E) All of the above have eight valence electrons.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would have to gain two electrons in order to achieve a noble gas electron configuration? O Sr Na Se Br
A) Br
B) Sr
C) Na
D) O, Se
E) Sr, O, Se
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Most transition metals do not form ions with a noble gas configuration.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which two bonds are most similar in polarity?
A) O-F and Cl-F
B) B-F and Cl-F
C) Al-Cl and I-Br
D) I-Br and Si-Cl
E) C-Cl and Be-Cl
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would have to lose three electrons in order to achieve a noble gas electron configuration? Si Mg Al Cl P
A) Si, P
B) Al
C) P
D) Cl
E) Mg, Al, P
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 147
Related Exams