Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which choice could provide the product. _______ + _______ 
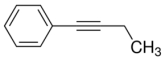
A) benzyl chloride,propyne
B) benzyl iodide,acetylene
C) phenyl bromide,1-butyne
D) phenyl bromide,2-butyne
E) phenyl iodide,ethene
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Reacting 2-nitrostyrene with iodobenzene under Heck-Mizoroki conditions will generate ____ as its product.
A) p-nitrostilbene
B) o-nitrostilbene
C) m-nitrostilbene
D) stilbene
E) p-aminostilbene
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the transition metal complex Pd(PPh3) 4.The total number of valence electrons this complex possesses is ____.
A) 2
B) 8
C) 10
D) 12
E) 18
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The third step in the mechanism of an alkene reacting with Wilkinson's catalyst is ____.
A) insertion
B) ligand association
C) oxidative addition
D) reductive elimination
E) ligand dissociation
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Transition metals can react with a variety of molecules called ____.
A) transition metal complexes
B) metals
C) ligands
D) complexes
E) covalent compounds
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
The valence electron count for Cr in CrC6H6(CO)3 is ____.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An 18 electron complex loses a ligand to become a 16 electron complex.This is an example of a(n) ____ reaction.
A) oxidative addition
B) ligand dissociation
C) insertion
D) ligand association
E) reductive elimination
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the third step in olefin metathesis the ____.
A) metal alkylidene ([M]=) undergoes a [2+2] pericyclic addition with an alkene alkene to form a metalcyclobutane intermediate
B) metalcyclobutane undergoes a reverse [2+2] addition (i.e.,retro [2+2] reaction) to generate a new metal alkylidene
C) reaction of the new metal alkylidene with a new alkene (by [2+2] addition) to generate a new metalcyclobutane intermediate
D) formation of the alkene product from a retro [2+2] reaction of the second metal alkylidene
E) alkene reacts with a second alkene by [2+2] addition
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Coupling a terminal alkyne with an alkenyl halide,using a mixture of a Pd and Cu(I) catalyst,is an example of ____.
A) Stille Coupling and Carbonylation reaction
B) Suzuki-Miyaura Coupling
C) Heck-Mizoroki Coupling
D) Sonogashira Coupling
E) Gilman Reagent-Mediated Coupling
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Coupling an aryl tin reagent with an alkenyl halide,using a Pd catalyst,is an example of ____.
A) Stille Coupling
B) Suzuki-Miyaura Coupling
C) Heck-Mizoroki Coupling
D) Sonogashira Coupling
E) Gilman Reagent-Mediated Coupling
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The first step in olefin metathesis has the metal alkylidene ([M]=) reacting with an alkene by ____ addition to form a ____ .
A) [2+2],metalcyclopropoane
B) [4+2],metalcyclobutane
C) [2+2],metalcyclobutane
D) [4+2],metalcyclopropane
E) [2+2],metalcyclopentane
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
A student wants to make 4-nitro-1-phenyl-1-butyne using Sonogahira coupling conditions,beginning with iodobenzene.This student will also require ____ in order to make this product.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the transition metal complex Ni(CN) 3(H2O) 23-.The total number of valence electrons this complex possesses is ____.
A) 2
B) 8
C) 10
D) 12
E) 18
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Sonogashira and ____ reagents both make use of copper reagents in order to perform cross-coupling reactions.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The fifth step in the mechanism of an alkene reacting with Wilkinson's catalyst is ____.
A) insertion
B) ligand association
C) oxidative addition
D) reductive elimination
E) ligand dissociation
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Converting an alkene to an alkane using the catalyst Pd/C is ____ hydrogenation.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following reaction.The complex in the middle underwent a(n) _____ reaction to make the final product. 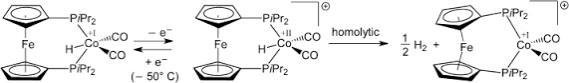
A) oxidative addition
B) ligand dissociation
C) insertion
D) ligand association
E) reductive elimination
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Reacting 1-pentyne with iodobenzene in the presence of CuI,Pd catalyst (in an amine base) will generate ____ as its product.
A) 3-phenyl-1-pentyne
B) 1-phenyl-1-pentyne
C) 1-phenyl-1-butyne
D) 1-phenyl-1-propyne
E) 3-phenyl-1-butyne
G) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
The total number of valence electrons contributed by all nitrogen atoms toward Re is ____. 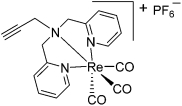
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 86
Related Exams