A) favor right side
B) favor left side
C) neither
E) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
What is the leveling effect?
Correct Answer

verified
It is the deprotonat...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
The tail of the curved arrow must start at ____ electron density and the head must end at ____ electron density.
A) low, high
B) high, high
C) low, low
D) high, low
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction, which reactant functions as an acid? 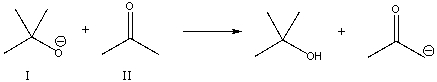
A) I
B) II
C) neither
E) None of the above
Correct Answer

verified
Correct Answer
verified
Essay
Draw the conjugate base of the following compound. 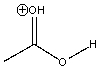
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the conjugate acid in the following reaction? 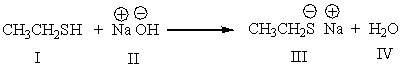
A) I
B) II
C) III
D) IV
E) Both I & IV
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction, identify the Lewis acid. 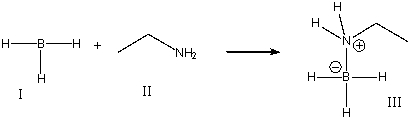
A) I
B) II
C) III
D) Both I & III
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Which compound has the most acidic proton? Explain your choice. 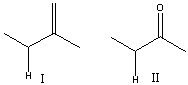
Correct Answer

verified
Compound II.
The conjugate base of both ...View Answer
Show Answer
Correct Answer
verified
The conjugate base of both ...
View Answer
Multiple Choice
The following compound is best classified as a ____. 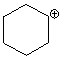
A) Brønsted-Lowry acid
B) Lewis acid
C) Brønsted-Lowry base
D) Lewis base
E) Both C & D
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is not a Lewis acid?
A) BF3
B) FeCl3
C) CH3CH2+
D) Ph3P
E) none of these
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Identify the acid and the base and provide a curved arrow mechanism for the following reaction. 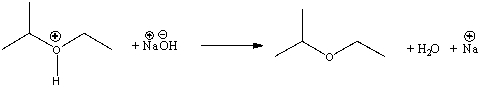
Correct Answer

verified
Correct Answer
verified
Essay
Aspartic acid, an amino acid, has the following structure. Circle the most acidic proton in aspartic acid. Explain your choice. 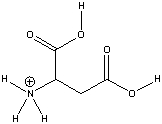
Correct Answer

verified
 The conjugate base ...
The conjugate base ...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
For the following reaction, which reactant functions as a base? 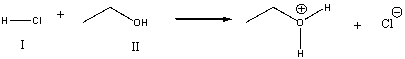
A) I
B) II
C) neither
E) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the strongest base that can be used with water as a solvent?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction, which reactant functions as a base? 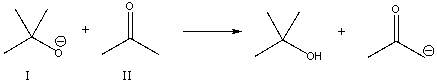
A) I
B) II
C) neither
E) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following protons in decreasing order (most to least) of acidity. 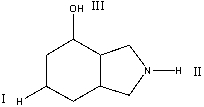
A) II>III>I
B) I>II>III
C) III>I>II
D) III>II>I
E) None of these
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction, identify the Lewis acid. 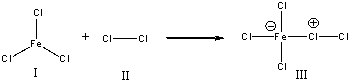
A) I
B) II
C) III
D) Both I & III
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction, which reactant functions as an acid? 
A) I
B) II
C) neither
E) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the counterion in (CH3) 3CLi?
A) CH3+
B) CH3-
C) C(CH3) 3-
D) Li+
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
For the following reaction mechanism, identify the error, if any and describe the modification necessary by drawing the correct mechanism. 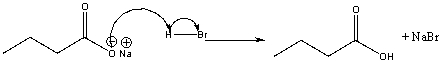
Correct Answer

verified
The first arrow from the lone pair of ox...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 61 - 80 of 126
Related Exams